ORONO, Maine — The University of Maine System (UMS) is on track to welcome students back to campus for the spring semester, UMS officials said Tuesday. Enhanced testing efforts, supported through federal funding, will allow UMS “to fulfill our educational and research missions this fall without being a source of outbreak or significant spread of COVID-19,” UMS Chancellor Dannel Malloy said in a release.
The Gov. Janet Mills administration is committing $8.15 million in federal Coronavirus Aid, Relief, and Economic Security (CARES) Act funding to reimburse UMS for its fall semester testing and personal protective equipment (PPE) expenses. Malloy said during a press conference Tuesday those expenses were close to $80 million.
“Governor Mills' commitment to reimburse the universities for first semester testing and PPE expenses helps provide Maine’s public universities with the resources we need to expand testing in the spring and stay on track for our students and for Maine,” Malloy said.
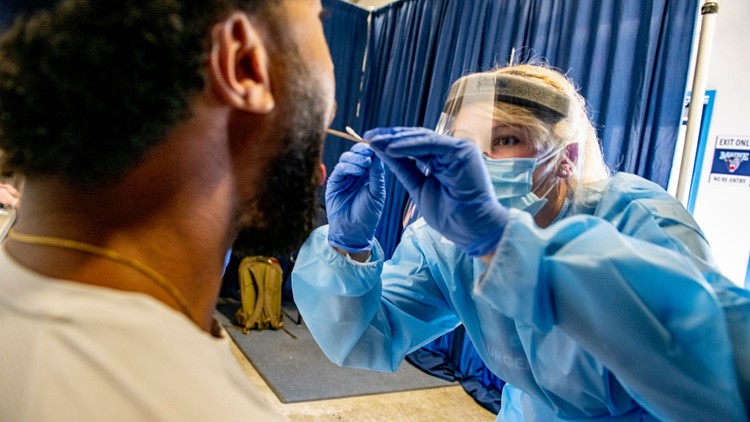
UMS will expand its testing program into the spring semester, supported in part by the state’s funding commitment. UMS conducted nearly 40,000 asymptomatic PCR COVID-19 tests during the fall semester. The University of Maine System, campus-based testing leaders, existing testing partners and other stakeholders are working on plans to expand testing and adapt screening and tracing strategies to increasing case counts.
“Shared commitment to our Together for Maine public health campaign and our screening strategies allowed us to fulfill our educational and research missions this fall without being a source of outbreak or significant spread of COVID-19,” Malloy said. “There is reason for optimism with the deployment of a safe and effective vaccine, but we expect to confront higher case counts in January than we did in August. We will respond with increased testing capacity.”
Resident halls will open and students will be welcomed back to campus with arrival screening the week of January 19 and a planned mix of in-persion, hybrid, and on-line classes beginning January 25. The individual universities will be distributing campus-specific details to students, faculty, and staff over the first few weeks of winter break.
Malloy appointed a task force on vaccine partnership and planning to expand and facilitate the use of university resources to assist with vaccine distribution, recommend needed changes to UMS immunization requirements, and contribute to student and public awareness about the efficacy and safety of FDA-approved vaccines.
The task force will brief the Board of Trustees Finance, Facilities, and Technology Committee at its Jan. 6, 2021 meeting and issue a report and work plan to the full Board on Jan. 25, 2021. The task force will be led by:
- University of Maine President Joan Ferrini-Mundy, Chair
- UMS Chief of Staff and General Counsel James Thelen
- UMS Chief Facilities Management and General Services Officer Chip Gavin
- UMS Executive Director of Public Affairs Dan Demeritt
UMS says the task force will engage with university scientists, nursing and public health program leaders, academic and student affairs leadership, communicators, and facilities and emergency response team members.
During the press conference Tuesday, Malloy said he wouldn’t consider making vaccines for COVID-19 mandatory for students and stuff unless they are fully approved, rather than for emergency use. Currently, the Pfizer and Moderna vaccines—which are now being administered to front line health care workers and long-term care residents and staff across the state under phase one of the state’s vaccination plan—were granted emergency use authorization by the FDA.
Under Maine’s vaccination plan, college-aged students in on- and off-campus settings are eligible to be vaccinated in the third phase. The general public, or anyone residing in Maine who didn’t receive the vaccine in a previous phase, will be eligible in the fourth and final stage of the plan.
UMS says it has been monitoring and planning for the distribution of vaccines for COVID-19 throughout the public health crisis. This has included providing input to various distribution plan discussions, offering state partners an inventory of 220 cubic feet of ultracold storage space available throughout the state to support vaccine distribution, and a survey of UMS nursing programs to identify individuals for surge staffing and other healthcare delivery opportunities.
“The most ambitious vaccination campaign in the history of the world is now underway,” Malloy said in a release. “FDA-approved vaccines for COVID-19 will end the pandemic and restore normalcy if enough people choose to be immunized. With lives in the balance, Maine’s public universities will be doing all we can to support the distribution of the vaccine and promote immunization as the smart and responsible choice for our students, faculty and staff, and Maine citizens.”
“Now is a critical time in the fight against COVID-19,” UMaine President Ferrini-Mundy, chair of the UMS Scientific Advisory Committee, said. “In record time, science has delivered a safe, effective vaccine for a disease that has infected millions and upended life across the planet. As challenging as 2020 has been, we can be proud that Maine has led one nation’s most successful, science-based public health campaigns in response to the pandemic. In the new year, Maine’s research university will be working with UMS institutions and public educators across the state to create awareness about the efficacy and safety of FDA-approved vaccines, and support an efficient and statewide distribution of the vaccine.”
Watch Tuesday's press conference here: